WHO-GMP Certified Manufacturing
State-of-the-art WHO-GMP certified manufacturing facility for Mebendazole production. International quality standards, advanced technology, and rigorous quality control for global pharmaceutical markets.
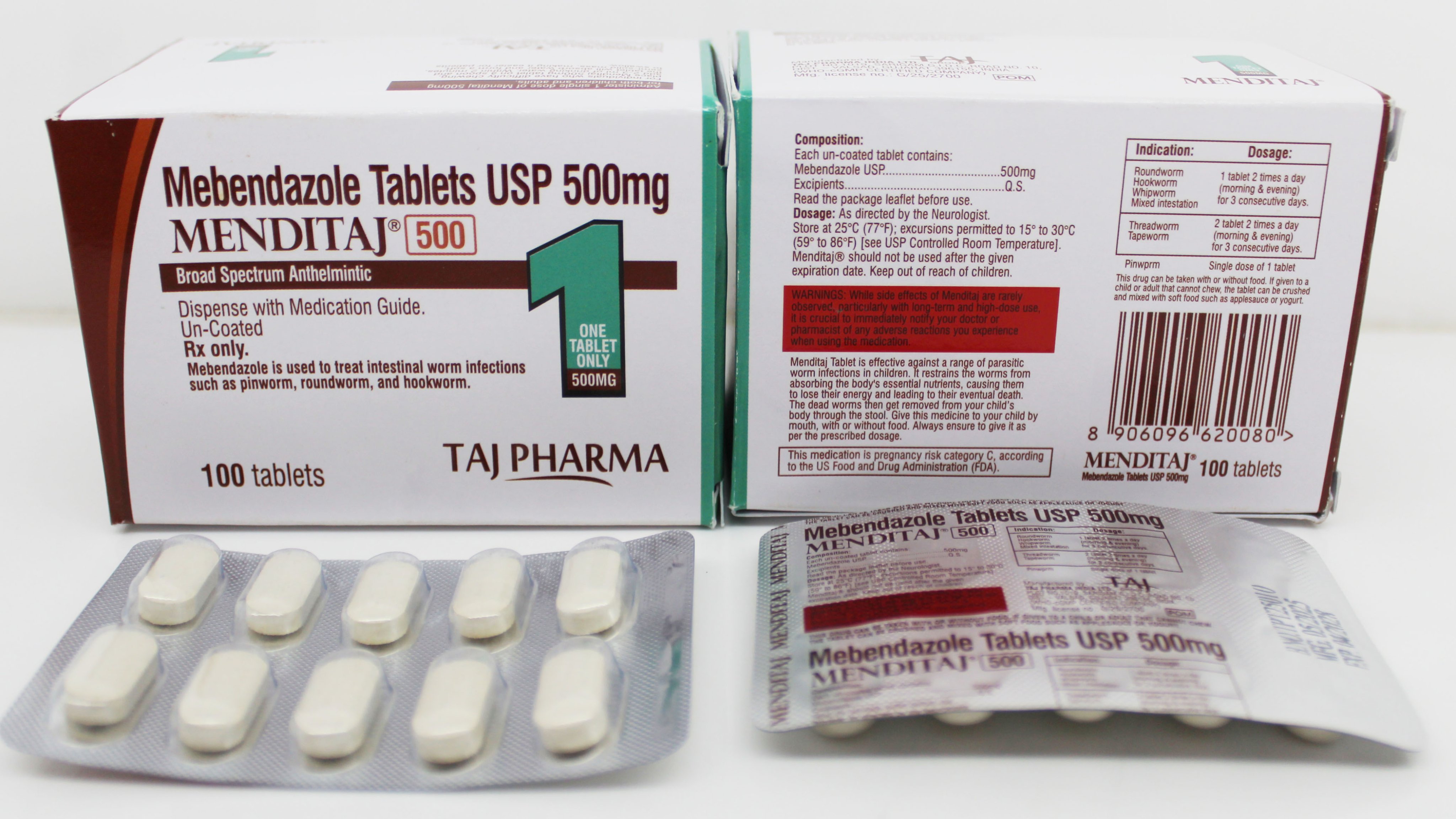
State-of-the-art WHO-GMP certified manufacturing facility for Mebendazole production. International quality standards, advanced technology, and rigorous quality control for global pharmaceutical markets.

Our WHO-GMP certified manufacturing facility represents the pinnacle of pharmaceutical quality standards. Designed and operated according to World Health Organization Good Manufacturing Practices, we ensure the highest quality Mebendazole production for global markets.
WHO-GMP Compliant
Tablets Monthly Capacity
Years Certification
Countries Exported
| Location: | Vapi, Gujarat, India |
| Total Area: | 100,000 sq. ft. |
| Clean Room Area: | 25,000 sq. ft. |
| Production Capacity: | 50 million tablets/month |
| Number of Lines: | 8 production lines |
| Automation Level: | 85% automated |
| QC Laboratory: | 10,000 sq. ft. |
| R&D Center: | 5,000 sq. ft. |
| Stability Chambers: | 20 chambers |
| Microbiology Lab: | 2,000 sq. ft. |
| Audit Trails: | Electronic system |
| Environmental Control: | HVAC system |
Comprehensive testing of all raw materials including API, excipients, and packaging materials according to pharmacopeial specifications.
Wet granulation process with precise control of particle size distribution and moisture content for optimal tablet compression.
High-speed tablet compression with real-time monitoring of weight, hardness, and thickness parameters.
Film coating process with uniform thickness and controlled release characteristics for specialized formulations.
Automated packaging lines for blister, bottle, and bulk packaging with integrated quality control checks.
Comprehensive final product testing including assay, dissolution, and stability before release.
| Assay: | HPLC method (98-102% of label claim) |
| Dissolution: | USP apparatus (85% in 30 min) |
| Hardness: | 5-8 kp (tablet hardness tester) |
| Friability: | <1% (friability tester) |
| Weight Variation: | ±5% (electronic balance) |
| Microbial Limits: | USP <61> microbial limits test |
| Air Quality: | HEPA filtered air, ISO 7 (Class 10,000) |
| Temperature: | 20-25°C ±2°C |
| Humidity: | 30-60% RH ±5% |
| Pressure Differential: | 10-15 Pa between zones |
| Air Changes: | 20-30 air changes/hour |
| Particle Monitoring: | Real-time particle counter |
| Process Validation: | 3 consecutive successful batches |
| Cleaning Validation: | Swab testing <10 ppm residue |
| Sterilization Validation: | Biological indicator testing |
| Computer Validation: | 21 CFR Part 11 compliance |
| Analytical Validation: | Method validation per ICH Q2 |
| Revalidation: | Annual revalidation schedule |
Comprehensive GMP training for all personnel with annual refresher courses and competency assessments.
Specialized technical training for equipment operation, analytical methods, and quality control procedures.
Occupational health and safety training including emergency response and PPE usage.
Training on SOP writing, batch record maintenance, and documentation practices.
Experience the highest standards of pharmaceutical manufacturing with our WHO-GMP certified facility. Quality, reliability, and compliance guaranteed for your Mebendazole requirements.
Complete Common Technical Document with Module 1-5 structure for regulatory submissions.
CTD DossierCertificate of Pharmaceutical Product for international trade and regulatory compliance.
COPP CertificateComprehensive stability data and shelf life information for all climate zones.
Stability DataOur team is ready to provide you with the best pharmaceutical products and support