Pharmacovigilance & Safety Monitoring
Comprehensive Mebendazole adverse event reporting system with pharmacovigilance guidelines, side effect monitoring, and safety reporting procedures for healthcare professionals and patients.
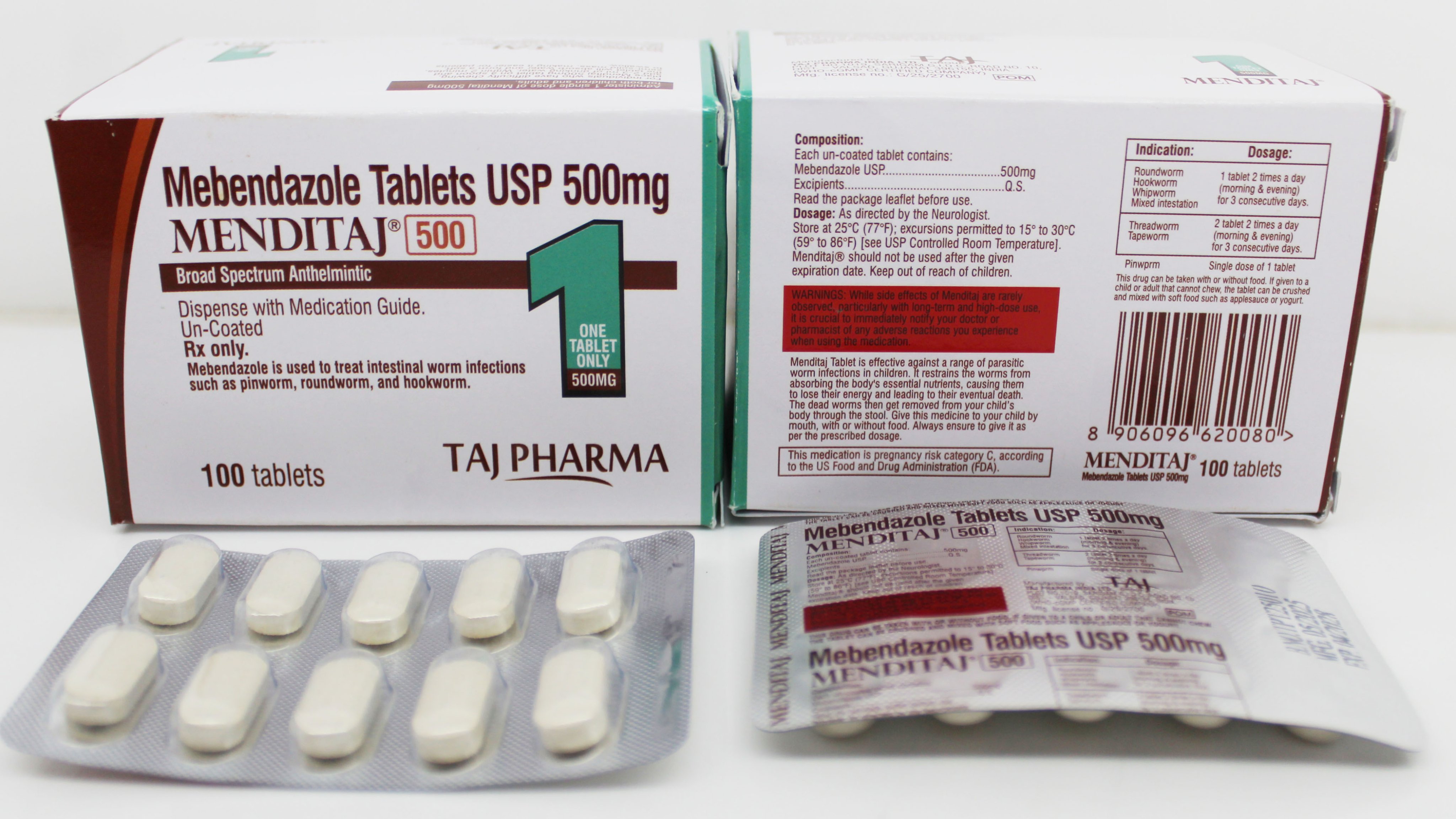
Comprehensive Mebendazole adverse event reporting system with pharmacovigilance guidelines, side effect monitoring, and safety reporting procedures for healthcare professionals and patients.

Pharmacovigilance is essential for monitoring the safety of Mebendazole and ensuring patient safety. This comprehensive guide provides healthcare professionals with the tools and procedures for effective adverse event reporting and monitoring.
Frequency: 2-5% of patients
Severity: Usually mild to moderate
Onset: Within 24 hours of administration
Duration: Typically 1-3 days
Management: Take with food, adequate hydration
Frequency: 1-3% of patients
Severity: Usually mild
Onset: Within 24-48 hours
Duration: 1-2 days
Management: Fluid replacement, avoid spicy foods
Frequency: 1-2% of patients
Severity: Usually mild
Onset: Within 24 hours
Duration: 1-2 days
Management: Take with food, rest
Frequency: <1% of patients
Severity: Usually mild
Onset: Within 24 hours
Duration: 1-2 days
Management: Rest, over-the-counter pain relievers
Frequency: <1% of patients
Severity: Usually mild
Onset: Within 24 hours
Duration: 1-2 days
Management: Avoid driving, rest
Frequency: <1% of patients
Severity: Usually mild
Onset: Within 24-48 hours
Duration: 2-3 days
Management: Rest, adequate sleep
Symptoms: Rash, hives, swelling of face/lips/tongue, difficulty breathing
Action: Seek immediate medical attention
Frequency: <0.1% of patients
Symptoms: Intense, persistent abdominal pain, vomiting
Action: Seek medical evaluation
Frequency: Very rare
Symptoms: Yellowing of skin/eyes, dark urine
Action: Immediate medical evaluation
Frequency: Very rare
Symptoms: Unexplained bruising, bleeding gums
Action: Seek immediate medical attention
Frequency: Extremely rare
| Form Type: | Healthcare Professional Adverse Event Report |
| Sections: | Patient info, drug details, event description, medical history |
| Time to Complete: | 15-20 minutes |
| Submission: | Online, fax, or email |
| Follow-up: | Required for serious events |
| Confidentiality: | Patient privacy protected |
| Form Type: | Patient Adverse Event Report |
| Sections: | Personal info, medication details, symptoms, timeline |
| Time to Complete: | 5-10 minutes |
| Submission: | Online, phone, or mobile app |
| Language: | Multiple languages available |
| Assistance: | Customer support available |
Comprehensive collection of adverse event data from multiple sources including healthcare professionals, patients, and clinical trials.
Rigorous analysis of safety data to identify signals, trends, and potential safety concerns.
Timely communication of safety information to healthcare professionals, regulatory authorities, and patients.
Ongoing improvement of safety monitoring systems based on feedback and emerging safety science.
| Spontaneous Reports: | Within 15 days of receipt |
| Serious Events: | Within 7 days (FDA), 15 days (EMA) |
| Periodic Reports: | Every 6 months (first 2 years), then annually |
| PSURs: | Every 6 months for first 2 years, then annually |
| Follow-up: | Required for serious and unexpected events |
| Documentation: | Maintain records for 15 years |
Intensive monitoring for new safety signals, establishment of baseline safety profile.
Regular safety review meetings, signal detection, and risk assessment activities.
Routine safety monitoring, periodic safety update reports, continuous risk-benefit assessment.
Long-term safety profile assessment, rare adverse event identification, benefit-risk evaluation.
Comprehensive guide to Mebendazole safety profile and adverse event management.
Educational videos on adverse event identification and reporting procedures.
Detailed guidelines for proper adverse event reporting and documentation.
Round-the-clock support for healthcare professionals with safety questions.
| Medical Affairs: | Expert consultation on complex cases |
| Pharmacovigilance Team: | Dedicated safety monitoring specialists |
| Regulatory Affairs: | Compliance and reporting support |
| Medical Information: | Product information and dosing guidance |
| Clinical Trials: | Study design and safety monitoring |
| Emergency Contact: | 24/7 emergency support line |
Your reports help us monitor the safety of Mebendazole and improve patient care. Report any adverse events promptly to ensure continuous safety monitoring.
Complete Mebendazole dosage guidelines for all age groups and infection types with safety considerations.
Dosage GuideComprehensive guide to intestinal worms, symptoms, and treatment options with safety information.
Worm TypesHealthcare professional resources including clinical guidelines and treatment protocols.
HCP ResourcesOur team is ready to provide you with the best pharmaceutical products and support